LipiGO® contains a completely natural extract from the yeast Saccharomyces cerevisiae (BGCC™) and acts in the gut without disturbing intestinal flora. It helps to reach a healthy weight and prevents from the rebound effect.
The 3 following claims are approved when marketing LipiGO® in the European Union:
- Treatment of Type I obesity and overweight
- Reduction of the rebound effect after a diet
- Reduction in the absorption of calories from dietary fat
LipiGO® contains β-glucan and chitin-chitosan, a patented mix of natural polysaccharides (BGCC) found in the Saccharomyces cerevisiae yeast cell wall.
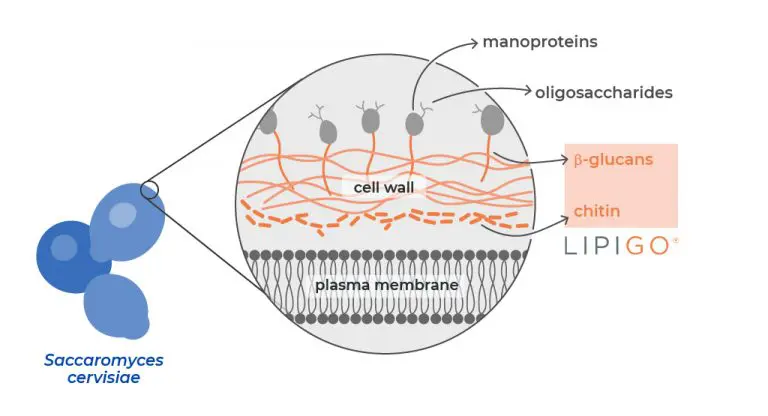
One feature that makes LipiGO® the ideal partner when tackling lifestyle and diet changes is its selective fat-binder activity for saturated fatty acids. Taking LipiGO® will not affect nutritional intake since it allows the absorption of other liposoluble compounds like vitamins and minerals.
Safety and efficacy has been clinically proven, demonstrating the products’ benefits after 12 weeks of treatment . These include 2 kg weight reduction without lifestyle changes, and 5 kg weight reduction with dieting. No adverse effects or toxicity were reported.
The product is patented (WO2014/001589) and was one of the 3 finalists of Nutraingredients Awards Asia 2020, awards that reward true innovation and cutting edge research in functional foods, dietary supplements and nutrition.
LipiGO® will be launched in the following months in several markets including China, European Union and Latin America countries.
Check LipiGO® clinical evidence here

